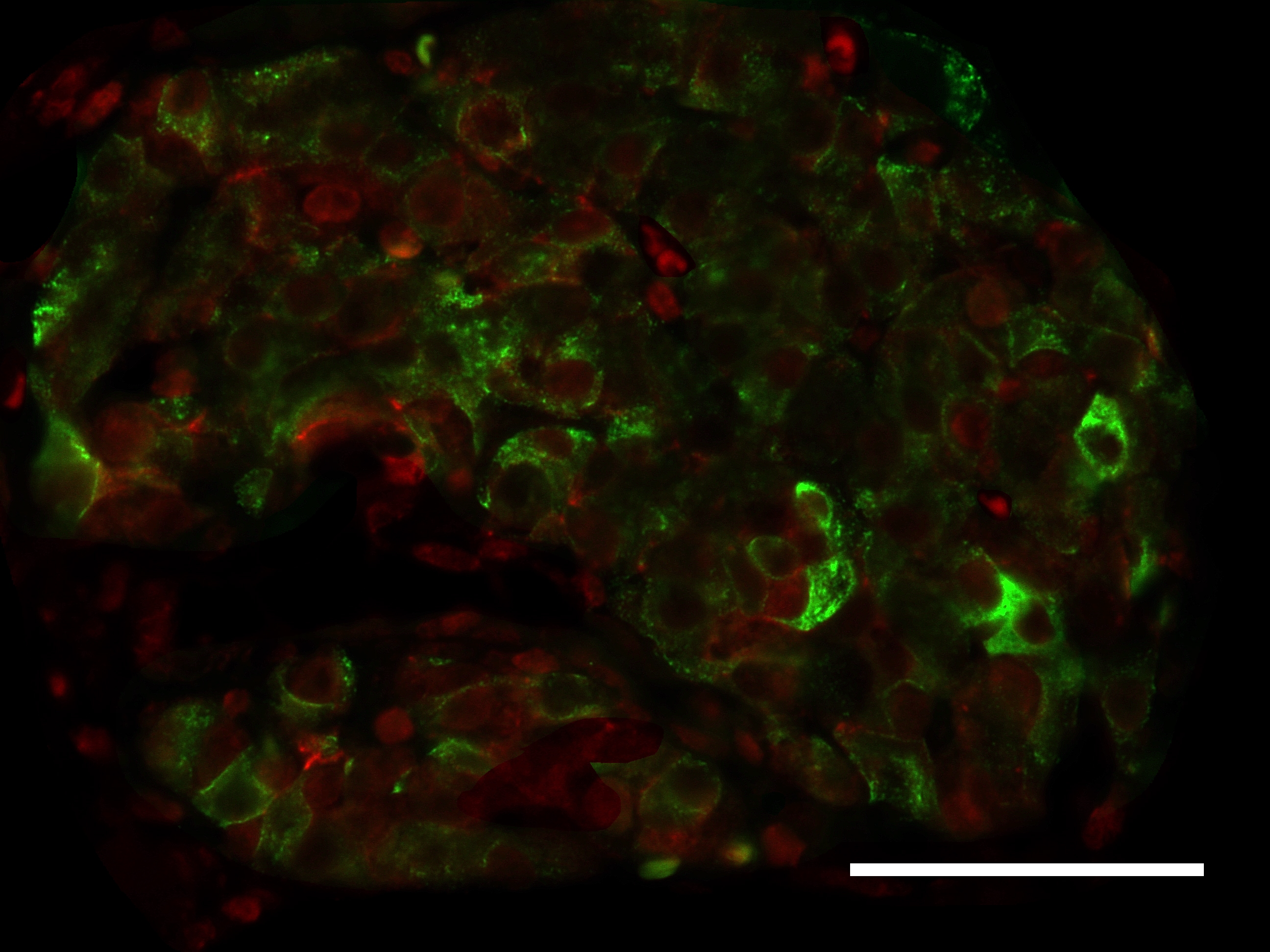
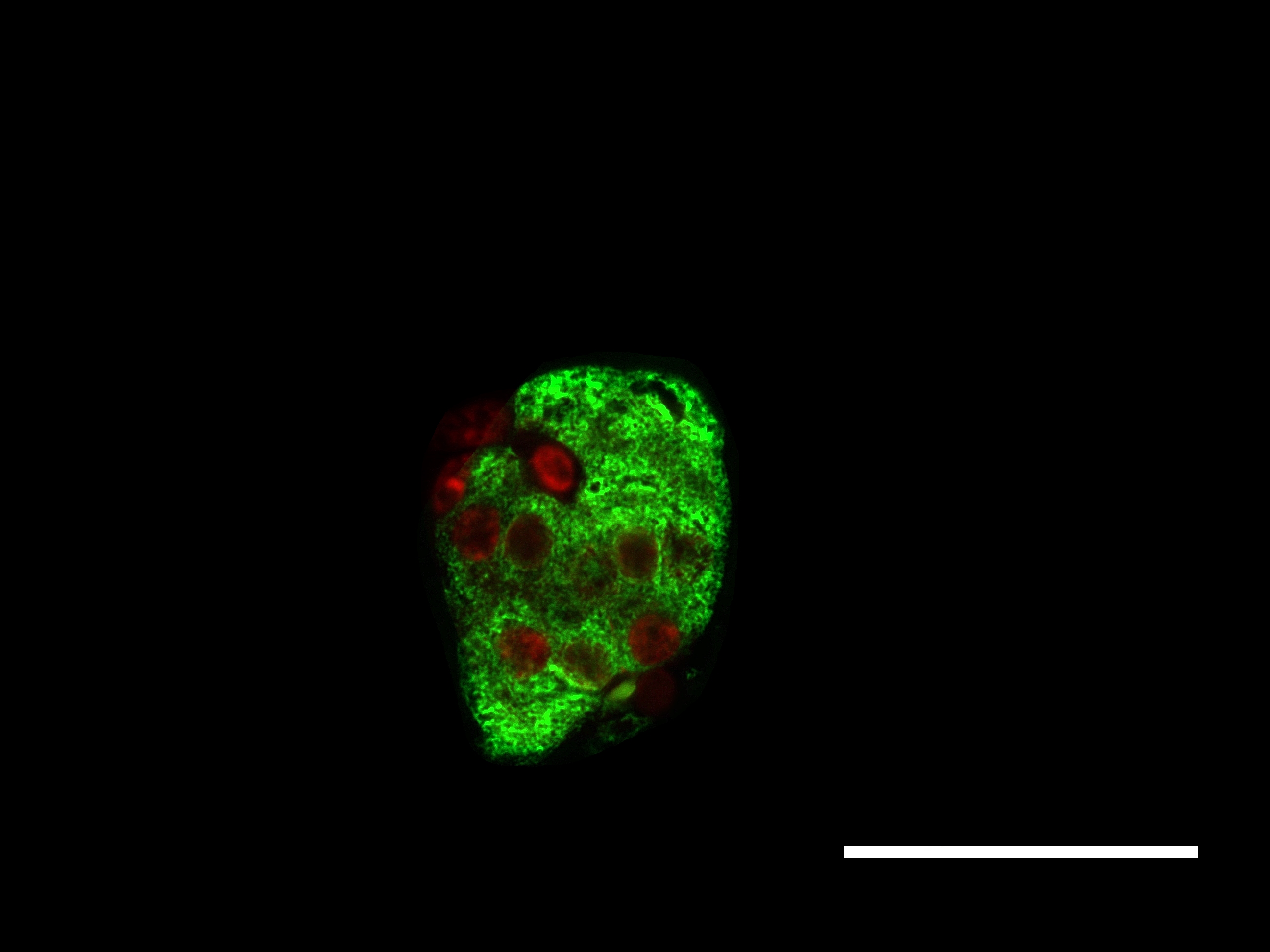
Glutaredoxin Systems
Type 2 diabetes mellitus (T2DM) is hallmarked by a progressive loss of beta-cells due to gluco- and lipotoxicity as well as a chronic state of inflammation [1]. Furthermore, oxidative stress and impaired redox signaling play a pivotal role in beta-cell decay [2]. There is also evidence for the occurrence of a hypoxa-like condition during high metabolic activity [3]. However, the islets of Langerhans broadly express thioredoxin family proteins [4]. This versatile family of proteins is a major actor in redox signaling and regulates distinct cellular pathways [5]. The Trx family comprises, among others, the glutaredoxin (Glrx) and thioredoxin (Trx) system. The Glrx system is mainly involved in redox regulation of cellular processes and biogenesis of iron-sulfur proteins. There is evidence that glutaredoxins (Glrxs) wield protective properties for the beta-cell [6-9] and that Glrx expression and activity are altered during T2DM [10, 11]. Mammals express four Glrxs classified as mono- or dithiol Glrx dependent on the number of cysteine residues in their active center. Dithiol oxidoreductases Glrx1 and 2 are major actors in thiol-disulfide exchange [12-14]. They exercise control over their targets by reversible post-translational de-glutathionylation of cysteine residues in dependence of glutathione reductase and glutathione. Monothiol Glrx3 and 5 are essential for biogenesis of proteins containing iron-sulfur-clusters [15, 16]. To elucidate the significance of the Glrx system for the beta-cell, we studied its expression in pancreatic islets of diabetic, obese homozygote db/db mice in comparison with heterozygote db/+ and C57Bl/6J wild type mice. Our findings revealed a significant loss in Glrx mRNA and protein expression in diabetic animals when compared to controls. Glrx-deficient islets presented a significant deprivation of their structural complexity, altered glutaredoxin distribution patterns, shifts in the cell cycle with increased apoptosis and diminished proliferation, loss of insulin content, and augmented production of reactive oxygen species (ROS). These findings correlated with the severity of the diabetic phenotype. Furthermore, we used beta-cell culture to simulate the diabetic metabolism in vitro. Concordantly, MIN6 cells exposed to lipotoxicity featured a loss of Glrx expression with impaired insulin secretion. Immunohistological Glrx5 (red) and insulin (green) staining of islets from 12 weeks old db/db (left) and C57Bl/6J mice (right), bars indicate 50 µm. After fixation and dehydration, sectioned pancreata were blocked and permeabilized. Afterwards, the slides were incubated with the primary antibodies followed by incubation of the secondary antibodies. Nuclei were stained with Hoechst and slides were preserved with ProLong Gold. There are two Trx systems in mammals: The cytosolic (Trx1 / TrxR1) and the mitochondrial (Trx2 / TrxR2) system, each including thioredoxin and its respective reductase. They are involved in regulation of DNA synthesis [17], gene expression [18], and proliferation [19] as well as apoptosis [20], but experimental evidence for a protective role of Trx is missing. Therefore, we have analyzed both Trx systems in hypoxic and cytokine-regulated stress using cultured beta-cells and isolated murine pancreatic islets. Protein levels of cytosolic Trx1 and TrxR1 significantly decreased, while mitochondrial Trx2 and TrxR2 were increased upon hypoxia exposure both in vitro and in vivo. We also used pig to mouse islet transplantation as a model of ischemia and reperfusion. When TrxR1/2 were chemically inhibited, survival of transplanted islets was substantially impaired. In contrast, extracellular Trx1 protected beta-cells from apoptosis and preserved glucose sensitivity in isolated murine pancreatic islets subjected to hypoxia. In conclusion, our data offers evidence for an essential role of both the Glrx and Trx systems in the survival of pancreatic beta-cells at diabetic conditions. Objectives: Literature: [1] R. P. Robertson, J. Harmon, Phuong Oanh Tran, Yoshito Tanaka, and Hiroki Takahashi. Glucose toxicity in beta-cells: type 2 diabetes, good radicals gone bad, and the glutathione connection. Diabetes., 52(3):581–7, Mar 2003. [2] R. P. Robertson, J. Harmon, P. O. Tran, and V. Poitout. Beta-cell glucose toxicity, lipotoxicity, and chronic oxidative stress in type 2 diabetes. Diabetes., 53:Suppl 1:S119–24, Feb 2004. [3] Y. Sato, H. Endo, H. Okuyama, T. Takeda, H. Iwahashi, A. Imagawa, K. Yamagata, I. Shimomura, and M. Inoue. Cellular hypoxia of pancreatic beta-cells due to high levels of oxygen consumption for insulin secretion in vitro. J Biol Chem., 286(14):12524–32, Apr 2011. [4] J. R. Godoy, M. Funke, W. Ackermann, P. Haunhorst, S. Oesteritz, F. Capani, H. P. Elsässer, and C. H. Lillig. Redox atlas of the mouse. Immunohistochemical detection of glutaredoxin-, peroxiredoxin-, and thioredoxin-family proteins in various tissues of the laboratory mouse. Biochim Biophys Acta., 1810(1):2–92, Jan 2011. [5] E. M. Hanschmann, J. R. Godoy, C. Berndt, C. Hudemann, and C. H. Lillig. Thioredoxins, glutaredoxins, and peroxiredoxins–molecular mechanisms and health significance: from cofactors to antioxidants to redox signaling. Antioxid Redox Signal., 19(13):1539–605, Nov 2013. [6] T. M. Reinbothe, R. Ivarsson, D. Q. Li, O. Niazi, X. Jing, E. Zhang, L. Stenson, U. Bryborn, and E Renström. Glutaredoxin-1 mediates NADPH-dependent stimulation of calcium-dependent insulin secretion. J Mol Endocrinol., 23(6):893–900, Jun 2009. [7] N. E. Ward, J. R. Stewart, C. G. Ioannides, and C. A. O’Brian. Oxidant-induced S-glutathiolation inactivates protein kinase C-alpha (PKC-alpha): a potential mechanism of PKC isozyme regulation. Biochemistry., 39(33):10319–29, Aug 2000. [8] N. Warwar, S. Efendic, C. G. Ostenson, E. P. Haber, E. Cerasi, and R. Nesher. Dynamics of glucose-induced localization of PKC isoenzymes in pancreatic beta-cells: diabetes-related changes in the GK rat. Diabetes., 55(3):590–9, Mar 2006. [9] Yosuke Watanabe, Colin E. Murdoch, Soichi Sano, Yasuo Ido, Markus M. Bachschmid, Richard A. Cohen, and Reiko Matsui. Glutathione adducts induced by ischemia and deletion of glutaredoxin-1 stabilize hif-1α and improve limb revascularization. Proc Natl Acad Sci U S A, 113(21):6011–6016, May 2016. [10] P. Di Simplicio, L. A. de Giorgio, E. Cardaioli, R. Lecis, M. Miceli, R. Rossi, R. Anichini, M. Mian, G. Seghieri, and F. Franconi. Glutathione, glutathione utilizing enzymes and thioltransferase in platelets of insulin-dependent diabetic patients: relation with platelet aggregation and with microangiopatic complications. Eur J Clin Invest., 25(9):665–9, Sep 1995. [11] Y. Du, H. Zhang, S. Montano, J. Hegestam, N. R. Ekberg, A. Holmgren, K. Brismar, and J. S. Ungerstedt. Plasma glutaredoxin activity in healthy subjects and patients with abnormal glucose levels or overt type 2 diabetes. Acta Diabetol., 9(2):225–32, Jul 2013. [12] A. Holmgren. Thioredoxin and glutaredoxin systems. J Biol Chem., 264(24):13963–6, Aug 1989. [13] C. A. Chrestensen, D. W. Starke, and J. J. Mieyal. Acute cadmium exposure inactivates thioltransferase (Glutaredoxin), inhibits intracellular reduction of protein-glutathionyl-mixed disulfides, and initiates apoptosis. J Biol Chem., 275(34):26556–65, Aug 2000. [14] S. A. Gravina and J. J. Mieyal. Thioltransferase is a specific glutathionyl mixed disulfide oxidoreductase. Biochemistry., 32(13):3368–76, Apr 1993. [15] R. Lill, R. Dutkiewicz, S. A. Freibert, T. Heidenreich, J. Mascarenhas, D. J. Netz, V. D. Paul, A. J. Pierik, N. Richter, M. Stümpfig, V. Srinivasan, O. Stehling, and U Mühlenhoff. The role of mitochondria and the CIA machinery in the maturation of cytosolic and nuclear iron-sulfur proteins. Eur J Cell Biol., 94(7-9):280–91, Jul-Sep 2015. [16] O. Stehling, C. Wilbrecht, and R. Lill. Mitochondrial iron-sulfur protein biogenesis and human disease. Biochimie., 100:61–77, May 2014. [17] T. C. Laurent, E. C. Moore, and P. Reichard. Enzymatic synthesis of deoxyribonucleotides. IV. ISOLATION AND CHARACTERIZATION OF THIOREDOXIN, THE HYDROGEN DONOR FROM ESCHERICHIA COLI B. J Biol Chem., 239:3436–44, Oct 1964. [18] C. Abate, L. Patel, F. J. 3rd Rauscher, and T. Curran. Redox regulation of fos and jun DNA-binding activity in vitro. Science., 4973(249):1157–61, Sep 1990. [19] G. Powis, D. L. Kirkpatrick, M. Angulo, and A. Baker. Thioredoxin redox control of cell growth and death and the effects of inhibitors. Chem Biol Interact., 111-112:23–34, Apr 1998. [20] M. Saitoh, H. Nishitoh, M. Fujii, K. Takeda, K. Tobiume, Y. Sawada, M. Kawabata, K. Miyazono, and H. Ichijo. Mammalian thioredoxin is a direct inhibitor of apoptosis signal-regulating kinase (ASK) 1. EMBO J., 17(9):2596–606, May 1998.